If you run a meat processing plant in the United States, you already know that the USDA’s Food Safety and Inspection Service (FSIS) requires every inspected facility to operate under a documented Hazard Analysis and Critical Control Point (HACCP) plan, no exceptions.
A HACCP plan for meat processing is your science-based blueprint for preventing, eliminating, or reducing biological, chemical, and physical hazards at every stage of production. According to the CDC, meat and poultry contribute to 22% of all domestically acquired foodborne illnesses and 29% of related deaths. These are the figures that underscore why a robust HACCP system is a public health necessity, not a bureaucratic formality.
In this guide, you’ll learn why a HACCP plan matters, how the principles translate into daily operations, what regulators expect, and how to build a plan that keeps your products safe and your auditors satisfied.
Why Meat Processing Needs a HACCP Plan
A HACCP plan for meat is the most effective framework for keeping contaminated products off store shelves. Here is why every processor needs one.
A Science-Based Preventive System
HACCP is a systematic, science-based process control system designed to prevent food safety hazards rather than detect them after the fact. Instead of pulling samples from finished batches, you identify where hazards are most likely to enter the process and stop them before they reach your products. This proactive approach has been the global standard for safe meat production since the early 1990s.
The Three Hazard Categories
Your hazard analysis must address three categories of risks that are common across meat processing plant operations:
- Physical hazards: Metal fragments, bone chips, glass, or plastic from packaging materials.
- Chemical hazards: Residual cleaning agents, excessive nitrite concentrations, or allergen cross-contact.
- Biological hazards: Pathogens such as Salmonella, E. coli O157:H7, Listeria monocytogenes, or Campylobacter.
Each category requires its own preventive measures, critical limits, and monitoring procedures. Skipping even one can expose your facility to recalls and consumer illness.
Regulatory Drivers
On July 25, 1996, the USDA FSIS issued its Pathogen Reduction/HACCP Systems Final Rule. It mandated sanitation SOPs, microbial testing, Salmonella performance standards, and HACCP programs for every inspected meat and poultry establishment. Implementation was phased: large plants (500+ employees) by January 1998, small plants (10–499 employees) by January 1999, and very small plants by January 2000.
The consequences of operating without a proper plan were put on full display in 2024 when a Listeria outbreak linked to deli meats sickened 61 people across 19 states and caused 10 deaths. Inspection reports revealed that food safety violations at the facility had been documented for at least two years prior.
Consumer Trust and Market Access
Beyond compliance, a well-documented HACCP plan for meat products signals to retailers and consumers that you take safety seriously. In an industry where a single recall can cost millions, your HACCP plan is both a regulatory requirement and a competitive asset.
Overview of the HACCP Principles
The seven core HACCP principles codified by the FDA’s National Advisory Committee on Microbiological Criteria for Foods (NACMCF) underpin every HACCP system:
- Conduct a Hazard Analysis: Using your process flow diagram, identify where biological, chemical, and physical hazards could reasonably occur and assess each for severity and likelihood.
- Identify Critical Control Points (CCPs): CCPs are steps where you can apply controls to prevent, eliminate, or reduce a hazard to a safe level. A CCP decision tree can guide your team, but expert judgment remains essential.
- Establish Critical Limits: Measurable boundaries separating safe from unsafe. For example, FDA guidance specifies cooking beef patties to 155 °F for 16 seconds.
- Establish Monitoring Procedures: Monitoring must be continuous or at defined intervals. Physical and chemical measurements are preferred over microbiological testing because they provide immediate results.
- Establish Corrective Actions: Pre-define what happens when a critical limit is breached. USDA requires you to identify the cause, restore control, prevent the affected product from entering commerce, and prevent recurrence.
- Establish Verification Procedures: Beyond monitoring, it includes calibrating instruments, conducting preshipment reviews, and reassessing the plan annually or whenever a process change occurs.
- Establish Record-Keeping Procedures: Detailed records, such as hazard analyses, CCP decisions, monitoring logs, corrective actions, and calibration data, are your proof of compliance. FSIS requires refrigerated-product records for one year and frozen-product records for two years.
These seven principles form an interlocking system: a weak hazard analysis leads to misidentified CCPs, irrelevant critical limits, and pointless monitoring. So, build each step on the one before it, and your plan becomes a self-reinforcing safety net.
Regulatory Requirements and Standards for Meat Processing
Knowing which regulations apply keeps you audit-ready year-round. Here is the regulatory landscape every meat processor must navigate.
USDA/FSIS Requirements
All meat and poultry products sold in the United States fall under the USDA FSIS HACCP program. Federally inspected facilities must maintain a validated HACCP plan, written SSOPs, and documented recall procedures. FSIS conducts daily on-site verification at slaughter plants and regular checks at processing facilities. A thorough understanding of USDA regulations for meat processing is critical before you begin writing your HACCP plan.
FDA Jurisdiction and Preventive Controls
Some meat products fall under FDA rather than USDA jurisdiction, particularly those containing less than a defined threshold of meat or poultry. These facilities must follow GMPs and risk-based preventive controls under 21 CFR Part 117 Subpart B. If your products cross jurisdictional lines, ensure you understand both FSMA compliance and USDA HACCP requirements.
Certification and Training
At least one person at every inspected plant must hold a recognized HACCP certification and be authorized to sign the plan and any revisions. HACCP training courses cover hazard analysis methodology, CCP determination, and documentation requirements. Without a certified team member on staff, your plant cannot legally operate under federal inspection.
International Standards
If you export, you’ll need to comply with Codex Alimentarius HACCP guidelines and potentially meet additional requirements such as EU hygiene regulations or certifications like SQF, ISO 22000, or BRC. Building your domestic plan with international standards in mind simplifies future market expansion.
Prerequisite Programs and Infrastructure for Building a HACCP Plan
Your HACCP plan depends on a solid foundation of prerequisite programs. These ensure that the basic conditions for safe production are already in place.
Good Manufacturing Practices (GMPs)
GMPs cover daily operational basics: personal hygiene, sanitation of food-contact surfaces, pest control, and preventive equipment maintenance. Without functioning GMPs, even the best HACCP plan will fail. Following core food safety practices consistently across shifts is non-negotiable.
Sanitation Standard Operating Procedures (SSOPs)
FSIS requires written SSOPs describing how your facility maintains sanitary conditions before, during, and after processing. It includes cleaning and sanitizing equipment, preventing cross-contamination between raw and RTE areas, and verifying water potability.
Employee Training and Standard Operating Procedures
Every employee who handles a product or monitors a CCP must understand their role in the safety system. Training should cover allergen control, proper thawing and cooking procedures, temperature monitoring, and corrective action protocols. Document all training sessions, these records become part of your regulatory file.
Facility Design and Workflow
Your plant layout should support sanitary processing with logical traffic flow from raw to finished product areas, adequate separation between species, properly designed drainage, and equipment that can be effectively cleaned. Temperature-controlled zones for receiving, storage, and processing are essential for cold-chain integrity.
Recordkeeping Foundation
Prerequisite program records, like cooler temperature logs, sanitation sheets, pest control reports, and calibration records, support your hazard analysis by showing that foundational hazards are under control. When an inspector asks why a hazard is “not likely,” these records are your evidence.
Developing a HACCP Plan for Meat Processing
Building your HACCP plan for meat processing follows a structured sequence. Here is how to move from concept to an inspection-ready document.
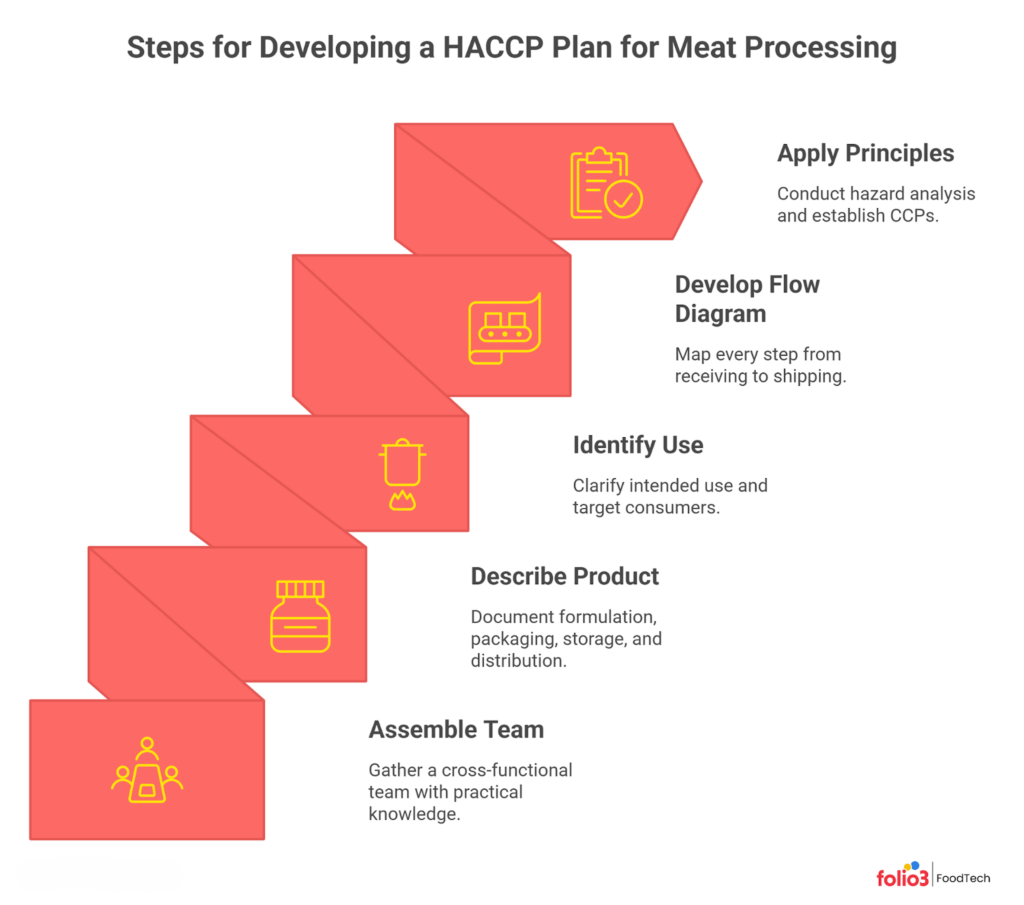
Assemble the HACCP Team
Assemble a cross-functional team that includes plant management, production-line supervisors, and quality assurance personnel. The people who make the product daily bring practical knowledge no outside expert can replicate, and FSIS expects hands-on understanding of every process.
Describe the Food and Its Distribution
For each product, document the formulation, packaging type, storage requirements, and distribution method. A ground beef product might use wax-coated paper or vacuum packaging, with shelf life from 4–14 days fresh to 6 months frozen. These details directly influence your hazard analysis because packaging and storage temperature determine which microbial hazards are relevant.
Identify Intended Use and Consumers
Clarify whether your product is ready-to-eat or requires cooking by the consumer. Ground beef is intended to be fully cooked by the end user, meaning the consumer’s cooking step is the final lethality point. RTE products must achieve pathogen destruction during your process, making in-plant CCPs even more critical.
Develop a Process Flow Diagram
Map every step from receiving raw materials through shipping. For ground beef, the flow includes receiving, storing, thawing, formulating, grinding, packaging, and freezing. For smoked sausage, add mixing, stuffing, cooking/smoking, chilling, and secondary packaging. Walk the production floor to verify your diagram matches reality.
Apply the Seven HACCP Principles
With your team assembled and the flow diagram verified, work through each principle for every product category. Conduct your hazard analysis, determine CCPs, set measurable critical limits, define monitoring and corrective action procedures, and establish verification and recordkeeping systems. The result is a living document that must be reassessed annually.
Example HACCP Plan for Meat Products
Seeing theory in practice makes the HACCP process concrete. Below is a simplified example of two common meat products that shows how hazard analysis and CCP identification work.
Ground beef: Contains beef (sometimes pork); intended to be fully cooked by the consumer; packaging includes wax-coated paper, freezer paper, and vacuum packs; shelf life 4–14 days fresh or 6 months frozen.
Smoked sausage/hot dogs: Fully cooked; intended to be reheated; vacuum-packed; shelf life approximately 2 months refrigerated or 6 months frozen.
Process Flow and Hazard Analysis
The table below summarizes key steps, hazards, preventive measures, and CCP assignments for a ground beef operation. Only selected steps are shown for brevity.
Receive Meat
- Potential hazards: Biological (E. coli O157:H7, pathogen growth), Physical (needles, shot), Chemical (none expected)
- Likely? No, suppliers provide validated interventions and letters of guarantee.
- CCP? No, hazard controlled at the supplier level.
Thaw Frozen Meat
- Potential hazards: Biological (pathogen growth during thaw)
- Likely? Yes, limit water temperature to 70 °F during thawing.
- CCP? No, controlled at a later processing step.
Formulate and Weigh Meat
- Potential hazards: Biological (pathogen growth, contamination), Physical (aluminum clips)
- Likely? Yes, maintain product temperature at 45 °F or below.
- CCP? Controlled later in the process.
Grind Meat
- Potential hazards: Biological (pathogen growth), Physical (bone chips), Chemical (cleaners/sanitizers)
- Likely? Yes, keep raw meat at ≤45 °F; use bone eliminator; follow sanitation SOPs.
- CCP? Yes, if the temperature could rise above the critical limit during grinding.
Package Product
- Potential hazards: Biological (pathogen contamination), Physical (metal fragments), Chemical (residual cleaners)
- CCP? Yes, when metal detection is the last barrier before distribution.
Interpreting the Hazard Analysis
Hazards deemed “not likely to occur,” such as BSE risk at receiving, controlled through supplier guarantees, do not require a CCP at that step. Pathogen growth becomes likely whenever meat warms above 40–45 °F, which is why temperature controls during thawing, grinding, and rework are critical. Physical hazards like metal or bone fragments require equipment-based interventions supported by regular maintenance and calibration records.
Lessons from Ground Beef and Smoked Sausage
These examples demonstrate that hazard analysis and CCP identification vary significantly by product type. Each product in your facility needs its own tailored plan, supported by scientific and regulatory documentation.
Ground beef: Because this is a raw product with no in-plant lethality step, CCPs focus on preventing pathogen growth through temperature control during processing and rework, and on ensuring packaging does not introduce physical hazards. Hazard likelihood assessments depend heavily on supplier documentation and historical testing data.
Smoked sausage: The process includes a cooking/smoking step (CCP-1) that provides lethality to destroy pathogens, and a chilling step (CCP-2) that prevents outgrowth of surviving organisms. Because lethality is achieved in-plant, receiving is not a CCP. However, mixing and weighing still require controls: use premixed cure to prevent excessive nitrite, and maintain meat temperatures at or below 45 °F.
Beef HACCP Plan for Meat Processing
Beef products are particularly susceptible to enteric pathogens such as Salmonella and E. coli O157:H7. Undercooked ground beef has been a recurring source of foodborne illness, making it one of the highest-risk categories in any meat processing facility.
Critical Limits for Cooked Beef Products
For cooked beef products like frozen beef patties, FDA guidance recommends an internal temperature of 155 °F for 16 seconds. Your HACCP team may set additional limits for oven temperature, belt speed, patty thickness, and humidity to ensure consistent lethality.
Raw Beef Controls
For raw ground beef, maintain product temperature below 45 °F throughout grinding, weighing, and packaging. Use supplier letters of guarantee to verify the absence of E. coli O157:H7 and BSE risks. These supplier assurances become a foundational part of your hazard analysis documentation.
Beef Jerky and Dry-Cured Products
For beef jerky, control hazards through marination pH, drying temperature, and water activity (aw). Critical limits must align with FSIS Appendix A (lethality) and Appendix B (stabilization) guidelines. These products present unique challenges because the preservation methods involve lower temperatures that may not achieve conventional cooking lethality.
Documentation and Records
Maintain comprehensive logs of cooking temperatures, cooling curves, grinding temperatures, and supplier certificates. If you use a commercial HACCP template, always customize it for your specific processes and products. A copy-paste approach will not satisfy an FSIS inspector.
HACCP Plans for Different Meat Products
Not all meat products carry the same food safety risks. Each product category demands its own tailored HACCP plan for meat products.
Processing Categories
FSIS requires separate HACCP plans for each processing category in your facility. These include:
- Raw ground products
- Raw not-ground products
- Thermally processed commercially sterile products
- Shelf-stable products (both heat-treated and not)
- Fully cooked not-shelf-stable products
- Heat-treated but not fully cooked products
- Products with secondary inhibitors
Pork and Poultry
Hazard profiles differ by species. Pork is associated with Trichinella, Yersinia, and Salmonella. Poultry hazards include Campylobacter and Salmonella, with significant cross-contamination risks. CCPs differ accordingly: poultry operations may designate scalding and chilling as CCPs, while pork sausage operations focus on the final cooking step.
Fermented and Cured Meats
Products like summer sausage or dry-cured ham rely on low water activity and pH to prevent pathogen growth. Monitor fermentation temperatures and track pH drop, typically to 4.6 or below within a defined time frame. Deviation can allow organisms like Staphylococcus aureus to produce toxins before stabilization.
Ready-to-Eat Deli Meats
Fully cooked meats require CCPs not just for cooking but also for post-lethality contamination. Maintain product temperatures at or below 40 °F after cooking, and consider post-packaging pasteurization as an additional safety barrier. Given the severity of Listeria risks in RTE meats, many processors also adopt environmental monitoring programs as a verification tool.
Integrating Supply Chain and Documentation into Your HACCP Plan
Your HACCP plan is only as strong as the ingredients entering your plant. Integrating supply chain controls and maintaining airtight documentation are essential for a plan that holds up under audit.
Supplier Programs
Hazards can originate long before raw materials reach your receiving dock. A formal supplier approval program means collecting supplier HACCP plans, validation data, certificates of analysis (COAs), allergen statements, and third-party audit reports. It also includes maintaining country of origin records, especially for ingredients with a documented history of specific hazards.
Letters of Guarantee
For raw meat inputs, supplier letters of guarantee demonstrate that specific hazards such as BSE materials or E. coli O157:H7 are controlled at the source. These documents become part of your HACCP file and directly support your hazard analysis when you determine a hazard is “not likely to occur” at receiving.
Logs and Records
Your HACCP records should include customer complaint logs, corrective action forms, training records, receiving logs, CCP monitoring data, calibration records, and shipping logs. FSIS requires slaughter and refrigerated-product records to be retained for at least one year, and frozen or shelf-stable product records for two years. All records must be on-site for a minimum of six months; after that, off-site storage is permitted if records can be retrieved within 24 hours.
As your operation grows, managing documentation manually becomes increasingly difficult. Optimizing your broader meat supply chain with integrated digital systems streamlines both supplier management and recordkeeping.
How to Digitize Your Meat Processing HACCP Plan with Foodtech
Imagine replacing manual temperature logs with IoT-enabled sensors that record cooler and cooking temperatures every few seconds, automatically flagging deviations from critical limits. Plus, cloud-based platforms store your hazard analyses, monitoring data, corrective action reports, and supplier certificates in a single searchable system with accessible to your team and FSIS inspectors on demand.
Digital audit software automates preshipment reviews, schedules annual reassessments, and generates compliance reports in clicks. The result is 24/7 audit-readiness: when an inspector arrives, your records are complete, organized, and instantly retrievable.
For processors managing complex operations, a purpose-built meat processing ERP can integrate HACCP monitoring with broader plant operations, connecting CCP data, traceability records, production scheduling, and quality management on a single platform. It makes managing a complex HACCP plan for meat processing virtually foolproof.
Conclusion
Developing a HACCP plan for meat processing is not optional, it is a federal requirement and the cornerstone of producing safe meat products. From assembling your HACCP team to conducting hazard analysis, identifying CCPs, setting critical limits, and building monitoring, corrective action, verification, and recordkeeping systems, every step matters.
Use the examples and frameworks in this guide as your starting point, but always adapt them to your specific processes and facility. No two plants are identical, and a HACCP plan that works for one operation will not necessarily satisfy another’s requirements.
A well-built HACCP plan protects public health, reduces your exposure to costly recalls, builds retailer and consumer trust, and opens doors to new markets. If you are ready to take action for building a digitized HACCP plan, connect with our footech experts.
FAQs
What Is the Difference Between a HACCP Plan and a Food Safety Plan?
A HACCP plan controls hazards at critical control points during processing. A food safety plan under FSMA is broader, covering preventive controls, supply-chain programs, and recall procedures. For USDA-inspected meat plants, the HACCP plan is the primary compliance document.
How Often Should You Reassess Your HACCP Plan?
FSIS requires annual reassessment at a minimum. You must also reassess when you change ingredients, processing methods, equipment, or packaging, or when an unforeseen hazard emerges. Treat your HACCP plan as a living document.
Can a Small Meat Processing Plant Use a Generic HACCP Template?
Generic templates are useful starting points, but every plant must customize to reflect its own products, processes, and layout. Inspectors verify that your plan matches actual operations, so a copy-paste approach will not pass.
What Happens if a Critical Limit Is Breached During Processing?
Execute your pre-defined corrective action immediately: identify the cause, restore CCP control, determine the disposition of affected product, and document everything. Failure to follow corrective action procedures can result in FSIS enforcement.
Do I Need Separate HACCP Plans for Each Product I Process?
You need a separate plan for each FSIS processing category. However, a single plan can cover multiple products within the same category, provided product-specific hazard differences are clearly documented.







