In 1984, more than 750 people in The Dalles, Oregon, fell ill after a deliberate salmonella contamination at local restaurant salad bars, the first documented act of bioterrorism through the U.S. food supply. The perpetrators were not outside hackers or foreign adversaries. They were people with access, intent, and opportunity.
That incident is a stark reminder that the greatest threats to your food supply do not always come from pathogens or poor handling. Sometimes, they come from people with a purpose.
This guide breaks down everything you need to know about food defense: what it is, why your facility needs a plan, how to build one from scratch, and what regulators expect from you in 2026. Whether you are a QA manager running a processing line or a food safety director overseeing a multi-site operation, this guide gives you a practical roadmap.
What Is Food Defense? Definition, Meaning, and Scope
Food defense is not food safety by another name. Understanding the distinction is the first step to building a system that actually works.
Food defense is the effort to protect food products from intentional adulteration or tampering by individuals seeking to cause public harm, economic disruption, or brand damage. Unlike accidental contamination, the defining factor in food defense is intent. Someone is trying to do this on purpose.
The FDA defines food defense as protecting the food supply from acts of intentional adulteration. FSSC 22000 expands this further, describing food defense as safeguarding food, ingredients, and packaging from intentional, malicious attacks.
The threats can be biological, chemical, physical, or radiological and they can come from inside your facility or outside it. The consequences of a single incident are severe. Think mass recalls, criminal investigations, loss of retail contracts, and the kind of brand damage that takes years to rebuild, if it gets rebuilt at all.
Food Defense vs. Food Safety vs. Food Fraud: What Is the Difference?
This is one of the most searched questions in the industry, and rightfully so. All three disciplines protect consumers, but each addresses a completely different root cause.
| Concept | Focus | Root Cause | Framework |
| Food Safety (HACCP) | Unintentional contamination | Pathogens, allergens, and natural hazards | HACCP / HARPC |
| Food Fraud (VACCP) | Economically motivated adulteration | Financial gain (e.g., diluting olive oil with cheaper substitutes) | VACCP |
| Food Defense (TACCP) | Malicious or ideological contamination | Intent to cause harm (e.g., disgruntled employee, terrorism) | TACCP / FSMA IA Rule |
These three systems overlap, but the intent behind the threat is what separates food defense from the others. And that difference is exactly why food defense requires its own standalone plan. Understanding food safety hazards is a strong starting point, but protecting against deliberate acts requires an entirely different lens. The Food Safety Suite from Folio3 integrates food defense procedures alongside your core food safety management system to ensure comprehensive protection.
Why Is a Food Defense System Needed? The Business and Regulatory Case
You might already have HACCP covered, your GFSI audit coming up, and your food safety manual polished. But none of that protects you from someone who wants to cause harm. Here is why a food defense system is not optional.
The Business Risks
A single intentional contamination event can cascade quickly. Here is what is at stake:
- Public health liability: Intentional contamination can affect large volumes of product before detection, putting hundreds or thousands of consumers at risk.
- Brand reputation: Consumer trust, once broken, rarely fully recovers. A contamination scandal follows a brand for years.
- Financial losses: Product recalls, legal costs, lost contracts, and operational shutdowns create significant financial exposure.
- Supply chain disruption: A contamination event at one facility can halt distribution networks across a region or country.
- Regulatory penalties: Non-compliance with the FSMA Intentional Adulteration Rule can result in facility shutdowns and enforcement actions.
The Regulatory Drivers
If the business case is not enough, the regulatory environment makes food defense mandatory for most covered facilities in the U.S. Here are the key frameworks you need to know:
FSMA Intentional Adulteration (IA) Rule — 21 CFR 121
The FDA’s FSMA IA Rule requires covered facilities to prepare and implement a written food defense plan, conduct a vulnerability assessment, identify actionable process steps (APS), and implement mitigation strategies. If you are a covered food facility, this is not a suggestion. Understanding your full FSMA compliance obligations is essential for any food manufacturer operating in today’s regulatory environment.
FSSC 22000 (Version 6)
FSSC 22000 requires organizations to conduct a documented threat assessment and implement mitigation measures integrated into the food safety management system. Food defense is treated separately from food fraud under this scheme.
GFSI Schemes: SQF and BRCGS
Both SQF and BRCGS align with GFSI food defense definitions and require food defense procedures as part of site certification. Even if FSMA does not cover you, your retailer may require GFSI certification, which means food defense requirements apply anyway.
PAS 96 and TACCP
PAS 96 and the TACCP (Threat Assessment Critical Control Points) methodology are widely used in the UK and internationally, and are recognized by FSSC 22000 as accepted approaches for conducting threat assessments.
A proactive food defense posture is also a competitive signal. Retailers, co-manufacturing partners, and global customers increasingly treat food defense maturity as part of supplier qualification. Implementing a food compliance management system demonstrates your commitment to regulatory excellence.
The Core Principles of Food Defense: What Every Program Is Built On
Before you write a single procedure, you need to understand the four pillars that every effective food defense program rests on. Think of these as the architecture behind your plan.
1. Deterrence
Make your facility a difficult, unattractive target. Deterrence is about visibility: access controls, CCTV, perimeter fencing, locked vats, clear signage, and a culture where employees know someone is paying attention. Most opportunistic threats will move on when they see a facility that takes security seriously.
2. Detection
You cannot rely on deterrence alone. Detection systems and protocols allow you to identify suspicious behavior or deviations before a threat escalates into an incident. This includes employee reporting protocols, security monitoring logs, supervisor checks, and anomaly detection in your process.
3. Response
When something goes wrong, your team needs to know exactly what to do and who to call. Written corrective action procedures, a clear escalation chain, and defined product hold protocols are the difference between a contained incident and a full-scale crisis.
4. Recovery
After an event, recovery involves more than cleaning up. It means conducting a root cause analysis, notifying the FDA and relevant authorities, executing a product withdrawal if needed, and rebuilding trust with your supply chain partners and consumers. A food recall management system can speed up product withdrawal execution and documentation when minutes matter most.
Inside Threats vs. Outside Threats
Most food defense frameworks differentiate between two categories of threat:
- Inside threats: Disgruntled employees, contractors, or temporary workers with legitimate facility access who choose to misuse it. Regulatory frameworks like the FSMA IA Rule treat insider threats as the higher-probability risk for most facilities.
- Outside threats: Terrorism, external sabotage, or economically motivated bad actors attempting to infiltrate the supply chain through deliveries, vendors, or unauthorized access.
Both require mitigation, but your vulnerability assessment will usually reveal that your greatest exposure is the person already holding a key card.
The FDA ALERT System: Food Defense Awareness in Action
The FDA developed the ALERT awareness tool as a practical, plant-level framework for food defense. It is particularly valuable for facilities of any size that want to embed food defense thinking into daily operations.
It is not a substitute for a full food defense plan under FSMA. But it is a habit-building framework that keeps your entire team from supervisors to line workers thinking about security every shift.
Breaking Down the A.L.E.R.T. Framework
A — Assure
Verify that every ingredient, raw material, and packaging component entering your facility comes from a known, trusted source. Require sealed containers and verified documentation for all incoming shipments. Reject any delivery with broken seals, inconsistent documentation, or a driver who cannot be confirmed against your approved roster. A robust supplier management system ensures every incoming material is verified and logged against your approved vendor list.
L — Look
Monitor and secure your facility at all times. It means functioning CCTV coverage of critical areas, controlled access points with enforced protocols, and secured air intakes, water systems, and chemical storage. Any unlocked door, propped-open emergency exit, or unfamiliar vehicle on site should trigger an immediate check and documented report.
E — Employees
Know who is inside your building. Enforce ID badge policies without exception. Background screenings should be standard for all new hires in sensitive roles. When an employee is terminated, immediate access revocation including badge deactivation and key retrieval. Visitors and contractors must be signed in, escorted, and accounted for at all times.
R — Reports
Accurate, dated security records are your paper trail when an incident occurs or an auditor arrives. Document access logs, visitor sign-ins, security deviations, and any unusual events during each shift. These records support corrective action follow-through and demonstrate good faith compliance with regulators.
T — Threat
Have a written response protocol in place before you ever face a credible threat. Your protocol should define who gets called first, how the product gets placed on hold, how you communicate with the FDA and law enforcement, and who speaks to the media. A threat is not the moment to figure out your response that should already be documented and rehearsed.
What Is a Food Defense Plan? (And How It Differs from a Food Defense Program)
These two terms are used all the time interchangeably, but they mean different things, and the distinction matters when you are building your system.
A food defense plan is a written document. It records your facility’s vulnerability assessment, identifies actionable process steps, mitigation strategies, monitoring procedures, corrective actions, and verification activities, as required under 21 CFR 121 of the FSMA IA Rule.
A food defense program is the ongoing system that brings the plan to life. It includes the culture, training, day-to-day execution, and continuous improvement activities that make your plan operational, not just a binder sitting on a shelf.
If you have the plan but not the program, you are likely to pass a desk audit and fail a real incident. Both are necessary.
The FDA’s Food Defense Plan Builder (Version 2.0) is a free, guided software tool that walks your team through vulnerability assessment, APS identification, mitigation strategies, and recordkeeping requirements. It is a strong starting point, particularly for facilities building their first plan.
How to Conduct a Food Defense Vulnerability Assessment: Step-by-Step
The vulnerability assessment is the backbone of your food defense plan. It is where you identify the specific points in your process that are most exposed to intentional adulteration, and set the priority order for everything that follows.
Here is how to build one that actually holds up under scrutiny.
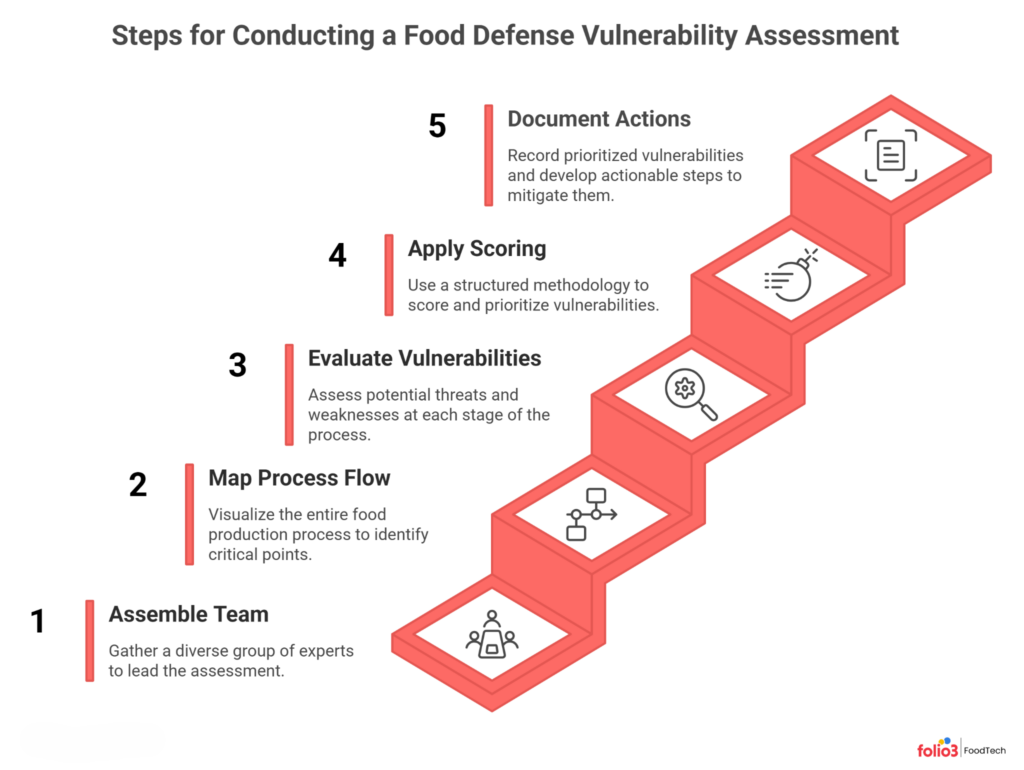
Step 1: Assemble Your Food Defense Team
A vulnerability assessment cannot be a one-person exercise. Pull together a cross-functional team that represents every part of your operation: QA, production, HR, maintenance, sanitation, purchasing, distribution, and security.
Appoint a Food Defense Coordinator, someone with the authority to enact policy changes and direct resources. The FSMA IA Rule requires that individuals performing food defense activities be qualified through relevant training, education, or experience. That includes your coordinator and the people carrying out monitoring and verification.
Step 2: Map Your Process Flow
Before you can identify vulnerabilities, you need to see your full process. Build a detailed process flow diagram that covers every step from raw material receipt to finished goods dispatch.
Mark every point where the product is open, accessible, or handled in bulk. Pay particular attention to:
- Bulk liquid receiving and tanker connections
- Open liquid storage tanks and holding vessels
- Secondary ingredient additions (open containers, manual additions to mixing vessels)
- Mixing, blending, and formulation steps
- Packaging lines and finished product staging
These are the areas where a small amount of contaminant can affect a large volume of product, and where detection is hardest before the product leaves your facility. Visibility into your full process also strengthens lot traceability which is essential if a withdrawal becomes necessary.
Step 3: Evaluate Vulnerabilities Using Key Activity Types
The FDA groups the highest-risk process steps into Key Activity Types (KATs). These are your starting points for evaluating vulnerability:
- Bulk liquid receiving and loading
- Liquid storage and holding
- Secondary ingredient handling
- Mixing, blending, and formulation
For each KAT and any other high-risk steps you identify, evaluate using the Three Elements of Risk:
- Potential public health impact: If adulteration occurred at this step, how many people could be affected? What volume of product would be contaminated?
- Degree of physical access: How accessible is the product at this point? Is it open, enclosed, monitored, or restricted?
- Attacker capability: Could a person realistically introduce a contaminant here without being detected?
Step 4: Apply a Scoring Methodology
The methodology you choose will depend on the size and complexity of your operation.
CARVER + Shock (quantitative)
CARVER + Shock scores each process step across six criteria: Criticality, Accessibility, Recuperability, Vulnerability, Effect, and Recognizability, plus a “Shock” factor that accounts for the psychological and economic impact of a contamination event. It is data-driven and systematic, making it well-suited for larger or more complex facilities. The resulting scores give you a ranked priority list of actionable process steps.
TACCP (qualitative)
Threat Assessment Critical Control Points (TACCP) is a team-based methodology that asks: who might attack, why, and how? It encourages cross-functional brainstorming and is recognized by FSSC 22000 as an accepted framework. It is particularly useful for organizations that want a structured conversation approach rather than a numeric scoring model.
FDA Vulnerability Assessment Tool (semi-quantitative)
The FDA’s free vulnerability assessment guidance is aligned with the IA Rule and produces a documented output showing which steps qualify as actionable process steps and why. It is the most compliance-ready option for U.S.-covered facilities.
The method matters less than the rigor. Whatever tool you use, document your reasoning for every step, including why steps were not identified as actionable.
Step 5: Document and Prioritize Actionable Process Steps
An actionable process step (APS) is a point in your process where mitigation is necessary to significantly minimize the risk of intentional adulteration.
For each APS, your documentation must include:
- The vulnerability identified
- Your scoring rationale (including all three elements of risk)
- Whether the step qualifies as an APS and why
- The mitigation strategy assigned to it
Prioritize based on the severity of public health impact first, then attacker accessibility. Steps with high public impact and low detection difficulty get addressed first.
Building Your Food Defense Program: From Plan to Practice
Once your assessment is complete, the work shifts from identifying vulnerabilities to building the operational system that keeps them controlled. This is where your plan becomes a program.
Developing Mitigation Strategies
For each actionable process step, you need at least one mitigation strategy that reduces the vulnerability to an acceptable level. Mitigation does not always mean expensive capital investment. Many of the most effective strategies are procedural.
Common mitigation strategies by category:
- Physical barriers: Locking lids on open vats, keycard-only access to mixing areas, locked hatches on liquid storage tanks, tamper-evident seals on bulk containers and finished goods.
- Personnel controls: Restricting access to specific zones based on job function, escort policies for contractors and visitors, and two-person verification for high-risk ingredient additions.
- Surveillance: CCTV coverage with daily footage review, motion sensors in restricted areas, and passcode-protected process controllers.
- Supplier controls: Sealed trailer requirements, verified documentation at receipt, inspection of incoming shipments before unloading, rejection of any shipment with broken seals or irregular documentation.
Match your mitigation strategy to the specific vulnerability at each APS. One size does not fit all.
Setting Up Monitoring Procedures
A mitigation strategy is only as good as the monitoring behind it. For each strategy, define:
- What is being monitored (e.g., hatch lock status on storage tank)
- How often it is checked (e.g., every 4 hours by shift supervisor)
- Who is responsible (by role, not just by name)
- How it is recorded (physical log, digital monitoring system)
Digital dashboards and automated sensor systems reduce human error and create a real-time audit trail. They also make it easier to spot patterns, repeated deviations at a specific step or shift that warrant investigation. A quality management system with monitoring dashboards delivers the real-time visibility your team needs to catch deviations before they escalate.
Corrective Actions and Verification
Corrective actions are what happen the moment a monitoring check finds a deviation. For example, if the hatch on a mixing tank is found unlocked, the corrective action is to secure the area, place the product on hold, review CCTV footage for the gap period, notify the Food Defense Coordinator, and investigate before releasing.
Corrective actions should be documented with the date, time, responsible person, root cause finding, and resolution.
Verification confirms the system is working as intended. It includes supervisor sign-offs, periodic internal audits, record reviews, and for more advanced programs, challenge testing.
Challenge testing involves running a controlled, scenario-based drill where an unauthorized person attempts to enter a sensitive area. You document the outcome, identify gaps, and update your plan accordingly. It is the food defense equivalent of a fire drill. Most competitors do not cover this, but it is one of the most effective ways to validate that your program actually works beyond the paperwork.
Training Frontline Staff
Your employees are your first and most important line of defense. Every person on the floor, in the warehouse, and at the loading dock plays a role.
Training should cover:
- How to recognize suspicious activity or behavior
- How to report a concern without fear of retaliation
- What the ALERT framework means in daily practice
- What to do if a delivery is suspicious or access is compromised
Keep training records. The IA Rule requires documented evidence of training activities. Use visual aids, scenario exercises, and refresher sessions at a minimum annually. Include contractors and temporary staff, insider threats do not always come from permanent employees.
Food Defense Plan Example: A Practical Table and Template Structure
This section makes the framework concrete. Here is a sample matrix showing how a real food defense plan documents a vulnerable process step from identification through corrective action.
Sample Food Defense Plan Matrix
| Process Step | Identified Vulnerability | Mitigation Strategy | Monitoring Procedure | Corrective Action |
| Bulk Liquid Receiving | Open tanker connection; unrestricted dock access | Sealed connections required; designated area; driver escorted at all times | Supervisor verifies seals and documentation at each delivery | Reject delivery; quarantine product; notify QA and Food Defense Coordinator |
| Liquid Storage Tank | Unlocked hatch; accessible to all plant staff | Keycard access only; hatch locked at all times | Shift supervisor checks hatch status every 4 hours | Stop production; secure area; review CCTV footage; notify QA; document findings |
| Secondary Ingredient Addition | Open containers in mixing area; limited oversight | Sealed containers until point of use; two-person verification for all additions | QA spot-checks ingredient addition log per shift | Hold batch; investigate discrepancy; document root cause; QA sign-off before release |
| Finished Product Packaging | Tamper risk at open packaging line | Tamper-evident seals; continuous CCTV coverage of line | The line supervisor verifies seal integrity each production run | Pull product; review camera footage; notify QA; conduct trace review |
Loading Dock Case Study
The loading dock is consistently one of the most commonly identified actionable process steps, and one of the most overlooked in day-to-day operations.
Assessment criteria for a typical loading dock: high criticality (large volumes of ingredients and finished goods move through), moderate to high accessibility (multiple personnel, vehicles, and contractors present), and high vulnerability (multiple handoff points with limited consistent oversight).
Mitigation strategies: keycard-only access to the dock area, dock doors closed when not actively in use, daily CCTV footage review, a visitor and driver sign-in log, and quarterly access audits.
Monitoring and verification: weekly access audits, daily footage spot-checks, monthly reassessments after any incident, and a semiannual training refresher for all receiving staff.
It is precisely the kind of scenario the FDA’s FSMA IA Rule is designed to address. Combined with strong food safety standards across the operation, a well-documented loading dock procedure becomes one of your strongest compliance and audit assets.
Conclusion: Build a Culture, Not Just a Compliance Document
A food defense plan gets you compliant. A food defense program built on training, vigilance, and continuous improvement is what actually protects your business and your consumers.
Compliance with FSMA and GFSI certification standards is the minimum bar. The facilities that lead in food defense go further: they run challenge tests, refresh their vulnerability assessments before the three-year deadline, involve every level of the workforce, and treat their food defense coordinator as a strategic role, not an administrative one.
In a market where retailers and global buyers are scrutinizing supply chain integrity more than ever, a mature food defense posture is a trust signal. It tells your partners that your operation is resilient, serious, and worthy of long-term business. Start or update your plan today! Contact our Foodtech experts to explore how a food safety management system can automate your food defense monitoring and compliance documentation.
FAQs
Who Is Required to Have a Food Defense Plan?
Under the FSMA Intentional Adulteration Rule (21 CFR 121), covered facilities that manufacture, process, pack, or hold food for U.S. human consumption must have a written food defense plan. Very small businesses and certain specific activities may qualify for exemptions. Review FDA guidance to confirm your facility’s coverage status.
How Often Should a Food Defense Vulnerability Assessment Be Updated?
The IA Rule requires reanalysis at least every three years, or sooner if a significant change occurs. A new process step, new equipment, new personnel, or a credible emerging threat all trigger an earlier review. Document your reanalysis date and findings every time as it is an audit requirement.
Is Food Defense the Same as HACCP?
No. HACCP addresses unintentional contamination from biological, chemical, and physical hazards and sits within your food safety management system. Food defense addresses intentional, malicious adulteration and is governed by the TACCP framework and FSMA IA Rule. They are separate systems with different triggers, but both should be integrated into your broader food safety approach.
What Is the CARVER + Shock Method in Food Defense?
CARVER + Shock is a quantitative vulnerability scoring tool that evaluates each process step across six criteria: Criticality, Accessibility, Recuperability, Vulnerability, Effect, and Recognizability, plus a Shock factor for psychological and economic impact. The output is a ranked list of your most exposed process steps, which then drives your mitigation priorities.
Can Small Food Businesses Skip a Food Defense Plan?
Some very small businesses are exempt from the FSMA IA Rule based on annual sales thresholds. However, GFSI certifications such as SQF and BRCGS require food defense procedures regardless of company size. And beyond compliance, any business serving the public carries a responsibility to protect the integrity of its products and its customers’ safety.
What Is the Difference Between a Food Defense Plan and a Food Defense Program?
A food defense plan is the written document: your assessments, mitigation strategies, monitoring procedures, and records. A food defense program is the ongoing execution: the culture, training, daily monitoring, corrective actions, and continuous improvement that make the plan operational every single day. You need both.







